UK 'head start' on EU means over-50s will be vaccinated by March, AstraZeneca chief says

The UK's "head start" in rolling out vaccines before the European Union means nearly everyone aged over 50 will be inoculated by March, the AstraZeneca chief executive has said.
Pascal Soriot said he believed the UK was on course to administer doses to "maybe 28 or 30 million people" within weeks – nearly half the total population – and would comfortably hit the target of vaccinating the most vulnerable groups by mid-February.
Watch: Michael Gove: UK’s vaccine supply is assured
It came amid rising international tension after the EU threatened to block vaccine doses from leaving the Continent without prior approval, leading Matt Hancock, the Health Secretary, to accuse Brussels of "protectionism and narrow nationalism".
Last week, the EU's rollout was thrown into disarray when AstraZeneca announced scheduled shipments of around 15 million doses would be delayed by manufacturing problems.
On Tuesday, EU politicians accused the firm of withholding the bloc's "fair share" of doses and suggested the UK had been given preferential treatment. But Mr Soriot said the EU had been "treated fairly," adding that Britain had a three-month "head start" after agreeing a deal for 100 million doses in May, three months before the EU.

He revealed that British plants had among the best yield in the world after spending months perfecting the process, while EU factories had been left with the "lowest productivity in the network".
"By March, the UK will have vaccinated maybe 28 or 30 million people. The Prime Minister has a goal to vaccinate 15 million people by mid-February, and they're already at 6.5 million. So they will get there," Mr Soriot told Italian newspaper La Repubblica.
Vaccinating at that rate would represent a significant acceleration, with the current NHS target to have given all 32 million over-50s their first dose by the end of April.
Mr Soriot also signalled that doses manufactured in the UK would not be diverted to the Continent after EU leaders reportedly asked whether British vaccines could help make up the shortfall.
"The contract with the UK was signed first and the UK, of course, said 'you supply us first' and this is fair enough," Mr Soriot said. "As soon as we can, we'll help the EU."
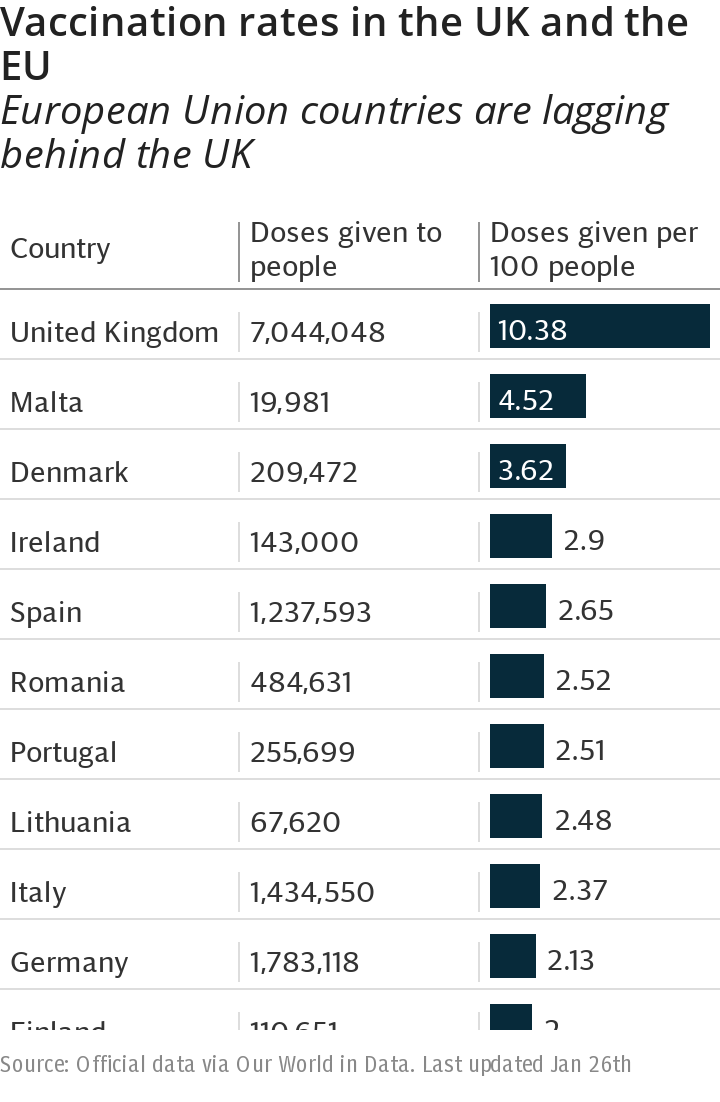
Sources at the firm also pointed out that Italy, Germany, France and the Netherlands had reached a preliminary agreement in June, only for the EU Commission to insist on formalising the deal – which took another two months.
Asked whether the EU had left it too late, Mr Soriot replied: "I will not pass judgment on this. But I can only tell you the facts, and the facts are that we basically signed an agreement with the UK three months before we did have it with Europe.
"The UK contract was signed three months before the European vaccine deal. So with the UK we have had an extra three months to fix all the glitches we experienced. As for Europe, we are three months behind in fixing those glitches.
Look, the sites that have the lowest productivity in the network are the sites that are supplying Europe. One of the plants with the highest yield is in the UK because it started earlier."
Sources close to AstraZeneca said dedicated supply chains had been set up for each international agreement to prevent countries squabbling over the same doses.
Whitehall sources said AstraZeneca’s three UK factories would be free to supply other countries, but only when 100 million doses of the Oxford vaccine had already been delivered to this country. "This is a commercial agreement. Those doses are for us, and they will not be going anywhere until all 100 million have been delivered to the UK," one Government source said.
It came amid increasing international acrimony, with Mr Hancock accusing the EU of "protectionism" by threatening to block doses from leaving Europe without prior approval.
Watch: Hancock - UK to help other countries identify Covid variants
EU leaders threatened legal action after AstraZeneca unexpectedly announced on Friday it would cut supplies in the first quarter of this year, a move that would reportedly mean a 60 per cent reduction to the agreed 31 million doses. Pfizer has also announced a temporary slowdown in deliveries of its vaccine while the firm upgrades its main factory.
Mr Soriot said that his firm had never made a contractual commitment to supply the doses, as Brussels has suggested. Instead, he said the company agreed to make its "best efforts" because the EU wanted to be supplied at more or less the same time as the UK, even though the contract was signed three months later. The best efforts falls short of a contractual commitment, which would weaken the EU's position were it to attempt to take legal action against AstraZeneca.
On a day of claim and counterclaim, the German government dismissed reports in the German press that the AstraZeneca vaccine was only eight per cent effective in over 65s.
However, on Tuesday night Der Spiegel reported that the German health minister was considering vaccinating younger people first amid concern that the Oxford vaccine had not been proven effective in older people.
Emer Cooke, head of the European Medicines Agency, refused to say what authorisation the EMA might give the AstraZeneca vaccine on Friday, but added it was “possible to conclude an authorisation that would focus on a particular age group or it's possible to conclude for a wider age group”.
Ursula von der Leyen, the European Commission president, said that the EU would press on with the export mechanism that would force companies to ask for permission before vaccines could leave the bloc.
In a speech to the World Economic Forum in Davos, Mrs von der Leyen said: “They must honour their obligations and this is why we will set up a vaccine export to transparency mechanism.”

UK officials were in talks with the EU to understand whether the move would impede supplies of the Belgian-made Pfizer vaccine, with Government sources insisting that the projected supply remained unchanged.
Mr Hancock said that ministers were "confident" that supplies would not be disrupted after conversations with the chief executives of both Pfizer and AstraZeneca.
He added: "But I would urge all international partners in fact to be collaborative and working closely together, and I think protectionism is not the right approach in the middle of a pandemic."
Diplomatic sources also claimed AstraZeneca vaccines meant for and paid for by the EU could have ended up in Britain, suggesting that the pharmaceutical company supplied the UK from the EU vaccine stock because Britain paid a higher price for the dose and approved it sooner.
However, Mr Soriot pointed out that the vaccine was being produced for no profit, and countries around the world had been charged effectively the same price.
"We want to treat Europe as best we can. You know, we do this at no profit, remember?" he said. "So we would like to treat Europe as good as possible. And I actually do believe we treated Europe fairly."
Watch: What UK government COVID-19 support is available?


